
an atom or ion with more than one electron. \(\sigma_r\), uncertainty in \(r\) from measuring \(r\). It is most widely used in the electrodynamics of charged particles, e.g. We are not permitting internet traffic to Byjus website from countries within European Union at this time. The experiment uses equations from circular motion and forces on electrons in magnetic fields which is part of the AS physics syllabus as well as being in the turning points in physics module. The charge to mass ratio of the electron is given by: e/m = 1.758820 1011 C/kg Where, m = mass of an electron in kg = 9.10938356 10-31 kilograms. reverse the orientation of the magnet? Which element is this? e/m, the ratio of charge of an electron to the mass of an electron. Since for a positively-charged particle, the helix is CW, the electron would move in a CCW helix if the magnetic field is moving towards us. 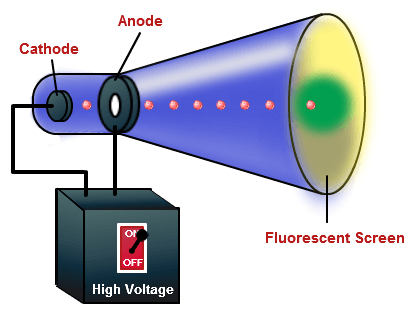
Start your trial now! The mass-to-charge ratio of the electron was first estimated by Arthur Schuster in 1890 by measuring the deflection of "cathode rays" due to a known magnetic field in a cathode ray tube. A vacuum tube with luminescent screen is used for both demonstrations. keep it constant while you vary the magnetic field by varying the What are the units used for the ideal gas law? 1 2 mv2 = eVacc Solving Eq. (9.7) for \(\sigma_B\) : Derive these expressions yourself verify that they are correct. electrons up to high velocity, and a magnetic field to then steer the If you recall from chemistry, every electron has an intrinsic magnetic moment-that's what gives it its "spin-up" (#m_s = 1/2#) or "spin-down" (#m_s = -1/2#) orientation. It is a more sophisticated field of theoretical physics which is concerned. In the visible region, higher is the energy, the, A: All things are provided in handwritten solution, A: Given: It is one of the fundamental constants of physics.It has a value of about 9.109 10 31 kilograms or about 5.486 10 4 daltons, which has an energy-equivalent of about 8.187 10 14 joules or about 0.511 MeV. respective uncertainties (measurement errors). The electron rest mass can be calculated from the Rydberg constant R and the fine-structure constant obtained through spectroscopic measurements. In blue light, conventional optical microscopes have a diffraction-limited resolution of about 200nm. Sign up to receive regular email alerts from Physical Review Letters. An electron beam can be used to supplement the treatment of areas that have been irradiated by X-rays. Modern Slavery Statement Note that older sources list charge-to-mass rather than the modern convention of mass-to-charge ratio. For an electron, the mass to charge ratio is: = |e| m = E B2r with e = 1.602 1019C being the elementary charge. 
If we alter the potential difference between the Y-plates, the beam is deflected upwards or downwards on the screen.Chemistry 1 Answer Truong-Son N. Usually, the potential difference applied to the X-plates makes the spot move across the screen at a uniform speed. 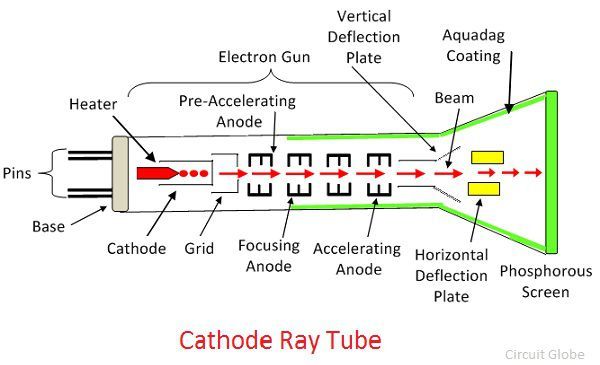
This deflection is produced by two pairs of parallel plates arranged at right angles. If necessary the stream of emerging electrons can be deflected in its passage between the gun and the screen. On leaving the gun, the electron stream passes across the tube and eventually hits the screen at the far side. Such arrangement of electrodes where a stream of electrons is produced is often known as the electron gun. As a result, the electrons accelerate across the gap between the electrodes and a narrow stream of the electrons emerges from the hole in the anode. A potential difference of some hundreds of volts is applied between cathode and anode. At a short distance from the cathode is an anode having a central hole in it. In the oscilloscope, the electrons are emitted by a hot cathode which is situated in a highly evacuated tube. The oscilloscope has many points in common with the discharge tube. The glow produced by the fast moving electrons on a fluorescent screen led to its use in radar and television. The discovery of cathode rays led to a vast field of practical application in Electronics. Modern cathode ray tubes have hot cathodes which require much less voltage, nearly 3000 volts. It needed a high voltage of the order of 30,000 volts. The cathode, in the discharge tubes used by Sir Thomson, was cold cathode. Experiments showed that mass of the electron is approximately 1/1860 of mass of hydrogen atom. Thomson found that the electrons had mass far less than of even the hydrogen atom. We know that hydrogen atom is the lightest atom. As these emanate from the cathode, the rays are called the Cathode Rays. These electrons emanate normally from the cathode. When this fluorescence was investigated, it was found that the fluorescence consisted of beams of negatively charged electrons. When the pressure in the discharge tube is less than 10 -4 mm of Hg, the discharge tube starts showing fluorescence.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
